Big news from The Joint Commission recently provided more motivation for better-performing hand-hygiene (HH) programs. Effective January 1, 2018, “. . . any observation by surveyors of individual failure to perform hand hygiene in the process of direct patient care will be cited as a deficiency resulting in a Requirement for Improvement (RFI) under the Infection Prevention and Control (IC) chapter for all accreditation programs.”1
The Joint Commission has more strong words regarding HH and HH programs. It states that, because “organizations have had since 2004 to implement successful hand hygiene programs, … there has been sufficient time for all organizations to train personnel who engage in direct patient care. While there are various causes for HAI [hospital-acquired infection], The Joint Commission has determined that failure to perform hand hygiene associated with direct care of patients should no longer be one of them.”1
Furthermore, “Surveyors also will continue surveying an organization’s hand hygiene program to National Patient Safety Goal (NPSG) 07.01.01.”1 The NPSG, introduced in 2004, requires healthcare organizations to (1) implement an HH program, (2) set goals for improving compliance with the program, (3) monitor the success of those plans, and (4) improve the results through appropriate actions.
If healthcare organizations need further reason to improve on and enforce their HH programs, the Joint Commission has provided it.
Better hand-care products encourage compliance
The first step in compliance to an HH program is cleaning the hands. Hands, particularly hands with damaged skin, present a maze of microscopic pathways where pathogens can hide and multiply. Painful skin damage from repeated hand cleansing is a major reason for non-compliance of HH protocols.
Ron Chappuis, Vice President of Healthcare Marketing, S.C. Johnson Professional (formerly DebMed), explained how their products encourage proper HH protocol, helping to break the cycle of damaged skin due to constant hand cleansing. “There is no other profession where hands are cleaned so frequently,” he said. “We fully recognize the compliance paradox that exists with products not formulated for high frequency use. The more inferior products are used, the more the skin is damaged, resulting in lower compliance. It is critical to ensure that the products being used are both mild and moisturizing to the skin.
from S.C. Johnson Professional
“S.C. Johnson Professional offers a portfolio of products specifically formulated for healthcare workers’ (HCWs) hands,” continued Chappuis. “Our products are blended with mild ingredients, to minimize damage, and they contain specially designed blends of emollients to provide added moisture to the hands. Each of our products contain unique aesthetic qualities that make each product pleasant to use, thus increasing the frequency of use.”
Chappuis highlighted the newest attribute of S.C. Johnson’s HH products, Invisi-Feel. “We formulated our newest foam sanitizer, Alcare Extra, to have virtually no residue. Healthcare workers notice the difference with just one use of the product. With Invisi-Feel, healthcare workers have nothing left on their hands except emollients.”
Important factors in addition to being mild and moisturizing include ease of use and effectiveness. If a product is not easily available, it is less likely to be used by HCWs. Ann Meyer, RN, BSN, Clinical Manager, Antiseptics, Mölnlycke, explained, “HIBICLENS has multiple features that encourage proper HH. It comes in an easy-to-use foam dispenser and has the lowest potential for skin irritation, when tested against seven other antiseptics (Mölnlycke 2017 irritation study). Additionally, noted Meyer, “HIBICLENS’ soap-based product will remove soil and debris, while the four percent chlorhexidine gluconate (CHG) begins killing germs on contact. HIBICLENS has a 24-hour residual effect on a wide range of germs.
“Clostridium difficile (C. diff) has been recognized as the most common HAI pathogen in acute-care hospitals,” continued Meyer. She referred to a study of HCWs’ hands sampled within half an hour of contact with patients who had C. diff, which found that 60 percent were contaminated, even following simple activities such as returning drug charts to the end of beds.2 “Compared to patients without C. diff infection (CDI), those infected experience an estimated 77 percent higher chance of being readmitted within 30 days and have a 55 percent longer hospital stay of nearly five days,” continued Meyer.
Relating the instance of a 689-bed hospital that experienced a 70 percent decrease in CDI rates with use of HIBICLENS for patient bathing, Meyer said, “It was concluded the C. diff spores may have been physically removed during the washing or bathing process, thus lowering the rates.3 HIBICLENS combines the action of soap-and-water washing with the advantage of CHG. CHG is active against vegetative cells and inhibits spore germination.”
Hand-hygiene monitoring systems enable accuracy
Direct observation, long considered the gold standard of HH monitoring, is time-consuming, labor-intensive, costly, and notoriously inaccurate. Electronic HH compliance monitoring ensures accuracy, transparency, and accountability.
“Hospital-acquired condition (HAC) scores now include methicillin-resistant Staphylococcus aureus (MRSA) and C. diff rates,” said Chappuis. “These two changes have resulted in more hospitals turning to electronic monitoring systems. While the key benefit of these systems is increased patient safety, hospitals are also better prepared for regulatory audits and now have accurate visibility to the top risk factor for HAIs: hand hygiene.
The DebMed System is clinically proven to be an accurate and reliable method of measuring compliance based on WHO 5-Moments and CDC guidance. He advised that the DebMed System offers robust real-time reporting capabilities, accessible online or by email, to analyze and interpret HH performance and trends for all unit personnel, including nurses, doctors, ancillary staff, patients, and visitors. It allows hospitals to compare how often its staff actually performs HH to how often they should perform HH, “allowing facilities to coach for improvement using reliable information,” Chappuis asserted.
A peer-reviewed study that measured HH compliance based on WHO 5 Moments showed a 746-bed teaching hospital’s HH compliance rate improved by 25.5 percent, accompanied by a reduction in hospital-onset MRSA infections by 42 percent and a reduction of costs by $434,000 over the study period.4
Kathleen Burzycki, Senior Marketing Manager, Ecolab Healthcare, also commented on the evolution of electronic HH compliance monitoring and talked about how their system encourages HH compliance. “More evidence is available today on the effectiveness of electronic HH compliance monitoring systems in reducing HAIs. We have noticed a significant uptick in the number of hospitals expressing an interest in this system, budgeting for it, and implementing it. The ability to demonstrate the impact of a compliance monitoring system on HH, infection rates, and patient safety in hospitals is key to implementation of this technology.
“We offer a programmatic approach,” said Burzycki. “Not only does this system monitor HH compliance but it also ties in sanitizer and soap, to offer hospitals a variety of efficacious options. Ecolab’s Compliance Monitoring System helps change HCW behavior by tracking patient interactions and dispenses, and by giving HCWs real-time feedback on their compliance in the form of lights and chirps. This can raise awareness and compliance immediately. The analogy we like to use is the seatbelt reminder light: when you forget to be safe, your car will beep and flash a light until you buckle up.
“We have tracked the Centers for Medicare and Medicaid Services’ rates of key infections, including MRSA, C. diff, catheter-associated urinary tract infection, and central-line–associated bloodstream infection, across five separate hospitals and have seen that, after one year of implementation, there has been a 25 percent decrease in these infections. This equates to nearly 100 fewer infections within a year,” stated Burzycki.
Burzycki pointed to a study on use of their system and corresponding increase in HH compliance.5 “By the end of the documented study period, three hospitals were averaging 86 to 90 percent compliance. These hospitals continue to utilize the system and continue to maintain and elevate these compliance rates.”5
Chris Hermann, PhD, Founder and CEO, Clean Hands – Safe Hands, talked about factors leading to the need for raising the bar on HH compliance. “Due to the growing pressures from insurers and regulatory bodies, HH is becoming a top priority for nearly all leading health systems.”
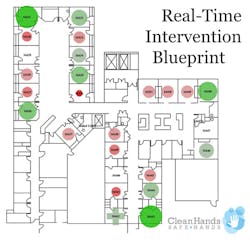
Hermann described how their reminder system takes HH performance to the next level in three ways. “First, our Real-Time Voice Reminder gently reminds providers to clean their hands when they forget. A human voice does the best job of getting their attention without alarm fatigue setting in. Hand hygiene typically doubles or even triples with the voice. Second, our data visualization suite offers unprecedented insights into the highest risk areas. Through our Performance Bubble Plots, managers can see individual clinician performance, to focus their efforts on the small handful of individuals that make up the greatest organizational risk. Our Real-Time Intervention Blueprints display HH performance by hospital room and patient condition, and risk-prediction algorithms identify whether there is a hot spot with poor HH and immediately alert leadership, so they can intervene before an HAI has a chance to spread. Third, our system is positive and fun rather than negative and punitive. Our patented six-phase Hand Hygiene Acceleration Pathway process is based on data-driven gamification, which keeps it fun. Customer surveys show a high level of staff acceptance.”
Hermann explained how their Adaptive Room Modes automatically adjusts their system to any different work flows, HH policies, or real-time interventions that may be needed. “For example, in a room with a C. diff patient, if a provider uses sanitizer prior to exiting, they won’t get credit for performing HH, and our Real-Time Voice Reminder changes to remind them to use ‘soap and water only.’
“Among hospitals that have installed the Clean Hands – Safe Hands system and followed our process for over six months,” continued Hermann, “100 percent have experienced a statistically significant decrease in HAIs by 45 to 81 percent.”
Ari Naim, President, CEO, and Co-Founder of CenTrak, also talked about recent regulatory changes as it relates to HH compliance measuring. “With the HAC Reduction Program, and more stringent changes made to The Joint Commission’s Infection Control Standards in January 2018,1 healthcare facilities are taking a closer look at their HH compliance monitoring methods. Expression of a zero-tolerance policy on improper HH by The Joint Commission is a step in the right direction.
“Automated and continuous monitoring is necessary to drive true compliance improvements and to intervene in real-time before the transmission of an HAI occurs,” observed Naim. “CenTrak’s electronic HH monitoring system measures true compliance of handwashing events throughout the entire facility, including bay-level locating technology in areas such as the post-anesthesia–care unit. Data can be viewed at the hospital, department, room, and individual level. Failure to obey HH protocols would result in a reminder from the staff member’s real-time location-enabled badge. Automated compliance reports allow facilities to reinforce training and intervene where needed most.” Naim added, “CenTrak’s system can be programmed to support custom protocols.”
Naim cited a study of three facilities using CenTrak’s monitoring system that concluded compliance rates were sustained at an average of 80 percent across all participating healthcare organizations, an increase from the average baseline of approximately 57 percent. “With over 80 months of data collection and more than five million HH opportunities evaluated, this monitoring system measured compliance on a group and individual level. CenTrak’s clinical-grade locating infrastructure, staff badges, and hand-washing monitors integrated directly into hygiene dispensers provided the most precise location data.6
SwipeSense CEO Mert Iseri offered a couple of reasons as to why many health systems are hesitant to adopt surveillance systems. “There are two major barriers to widespread adoption by hospital executives when it comes to adopting electronic HH monitoring systems. They must understand and accept that (1) the compliance rates they have been reporting for decades are inaccurate and (2) the small sample size—on average, observing 0.1 percent of the actual events—does not adequately portray the reality of what’s happening in their hospitals.”
Tom Derrick, co-founder of OpenMarkets, stressed the need for a system that can track individual HH performance, a need that has grown in importance since The Joint Commission statement.1 “Our data show SwipeSense as the fastest growing HH surveillance system on the market,” said Derrick. “But, given the strong outcomes, health systems are still adopting this game-changing technology slower than expected.”
“There is confusion in the marketplace regarding systems that provide usage statistics versus individual compliance. Simply having electronics on a dispenser to track the amount of gel or soap dispensed does not mean it is designed for individual accountability. The software matters more than the hardware,” continued Derrick. “Hospital leadership needs to evaluate the systems based on which system makes it simple for all clinicians to access and understand their compliance score.”
Derrick said that SwipeSense provides robust data on individual compliance as well as robust stratified reports on overall compliance. He related the experience of one SwipeSense user: “Outcomes data from SwipeSense usage at MacNeal Hospital in Chicago demonstrate both positive behavior change and a lowered risk of HAIs. Over a 12-month time frame, HH compliance in the areas using SwipeSense were 194 percent higher at MacNeal than in the areas not using the system.”
Charles Bareis, MD, Chief Medical Officer at Tenet Health Midwest, commented, “When we started getting data from SwipeSense, it was painfully startling, Our reports had shown compliance in the 90s but it was actually in the 20s. SwipeSense gave us the individual-level data we needed to encourage more diligent behavior.”
References
- The Joint Commission. Accreditation and Certification. Effective Jan. 1, 2018: individual hand hygiene failures to be cited under IC, NPSG standards. https://www.jointcommission.org/issues/article.aspx?Article=IlZJaLJCiRBZC2IRvnKkJTqEEU2n1Rxv3fqmsKqKPb0%3d&j=3545832&e#.WoYL4cTvozM.email. Last accessed August 31, 2018.
- Association for Professionals in Infection Control and Epidemiology, Inc. APIC Implementation Guide. APIC guide to preventing Clostridium difficile infections. https://apic.org/Resource_/EliminationGuideForm/59397fc6-3f90-43d1-9325-e8be75d86888/File/2013CDiffFinal.pdf. Last accessed August 31, 2018.
- Rupp ME, Cavalieri RJ, Lyden E, et al. Effect of hospital-wide chlorhexidine patient bathing on healthcare-associated infections. Infect Control Hosp Epidemiol. 2012 Nov; 33(11): 1094-1000.
- Kelly JW, Blackhurst D, McAtee W, et al. Electronic hand hygiene monitoring as a tool for reducing health care-associated methicillin-resistant Staphylococcus aureus infection. Am J Infect Control. 2016;44:956-957.
- Edmisten C, Hall C, Kernizan L, et al. Implementing an electronic hand hygiene monitoring system: lessons learned from community hospitals. Am J Infect Control. 2017 Aug;45(8):860-865.
- Buckner J, Read M. Individual monitoring increases hand-hygiene compliance in multicenter registry utilizing badge-based locating technology. Am J Infect Control. 2016 Jun;44(6):Suppl S94. https://www.ajicjournal.org/article/S0196-6553(16)30130-4/abstract. Last accessed August 30, 2018.
About the Author

Susan Cantrell
Susan Cantrell is Infection Prevention Editor for Healthcare Purchasing News.