If it isn’t clean it isn’t sterile. Luckily, there are a number of effective guidelines, practices and products available to support successful decontamination. New guidance documents are also on the horizon that will impact decontamination processes in central sterile/sterile processing (CS/SPD) departments. Experts weigh in on what to expect. We’ll also take a look at some of the products and solutions CS/SPD staff can use when decontaminating a variety of surgical devices.
New guidance documents
During the International Association of Healthcare Central Service Materiel Management (IAHCSMM) 2017 Annual Conference & Expo, May 7-10, 2017, in Nashville, attendees learned about two new documents that have the potential to change their decontamination practices:
- The new AAMI ST90: Processing of health care products Quality management systems for processing in health care facilities
- Updates to ANSI/AAMI ST79 Comprehensive guide to steam sterilization and sterility assurance in health care facilities
Richard W. Schule, MBA, BS, FAST, CST, FCS, CRCST, CHMMC, CIS, CHL, AGTS, ASQ CQIA, Director Clinical Education, STERIS Corporation, served as co-chair along with Damien Berg, Sterile Processing Manager at St. Anthony’s Hospital, Lakewood CO, on the committee that developed the new AAMI ST90. He comments on this new document, as well as the updated ANSI/AAMI ST79, which are both slated for release this summer.
“AAMI ST90 is a different twist on processing devices, in the sense that this document helps to establish a blue print or framework for establishing quality management systems for CS/SPD professionals assisting the coordination of all the silos of communication currently taking place,” said Schule. “I like to refer to AAMI ST90 as the connectivity that will bring together all of the ingredients that go into device processing as we need a more holistic approach to the functional definition of quality.”
According to Schule, AAMI ST90 identifies with several normative references, one of them being ANSI/AAMI/ISO 13485 Medical devices—Quality management systems— Requirements for regulatory purposes and the working group’s vision was to design a CS/SPD equivalent to the medical device manufacturing industry, which specifies requirements for a quality management system. He explains how the new AAMI ST90 presents the opportunity for CS/SPD to reevaluate and define the department’s scope of work, reexamine processes, define terms and definitions and “dial further into the principles of quality management.”
“Additionally, the new AAMI ST90 covers management responsibilities, resource management, and measurement, analysis and improvement. In most cases, CS/SPD around the country are probably achieving 65 to 75 percent of what will be required,” said Schule. “Documentation requirements will be a little more detailed than in the past, and there will be a greater commitment to training, education and competency, as well as process verification. We are excited for the opportunities this document will afford those responsible for device processing in their health care facilities.”
Though the draft is not finalized, the healthcare industry could see a summer 2017 update to ANSI/AAMI ST79. Schule notes the document has grown to include a few more definitions, an update to environmental requirements including instrument air, as well as ergonomic specifications and support of resources necessary to perform various processes. Verification of processes has also been enhanced to provide better guidance. Product realization to include product labeling and traceability will be further defined.
“Overall, a lot of hard work by Working Group 40 and the guidance of the document co-chairs, Ramona Connor and Cynthia Spry went into this enhanced document and every device processing department should have a copy on its reference shelf to support their department’s standard of work, work instructions and policies and procedures as well as a comprehensive education, training and competency program,” said Schule.
New products and solutions for decontamination
When speaking with CS/SPD professionals, a common complaint is that they don’t have the products and equipment they need to comply with industry standards. In this section, we cover some common challenges in decontamination, and the products that help overcome these issues.
Problems with pre-cleaning
“One challenge that CS/SPD staffs often face in order to achieve the successful decontamination of surgical instruments is the ability to receive instruments from the operating room (OR) in a timely manner,” said Todd Campbell, President of TBJ. “If surgical instrumentation is not received from the OR in an efficient manner after completion of a surgical procedure, then blood, body fluids and tissue will dry on the instruments making the technicians job of manually pre-cleaning instruments much more difficult and time consuming. Ideally, instruments should be rinsed under water immediately after surgery. If this isn’t possible, a moist towel should be used to cover the instruments to help prevent blood and other material from drying on the instruments before they reach the CS/SPD for processing to promote more efficient pre-cleaning by the CS/SPD staff.”
TBJ’s new Hydro-Force rinse system greatly assists technicians in the pre-cleaning and initial removal of gross debris of material from instruments after surgery. The Hydro-Force rinse system option enables technicians to place grossly soiled trays of instruments in the company’s pre-cleaning sinks and let the sink do the work for them. Water is automatically recirculated in the sink basin to create a gentle turbulence and remove gross soil and debris for a pre-programmed amount of time at the push of a button. Additional features include automatic sink basin filling, detergent injection and automatic draining. TBJ’s single basin units can be installed in the OR area to be used for immediate, automated rinsing of instruments before instruments are sent to CS/SPD. The Hydro-Force option can also be included in TBJ’s triple basin sinks designed specifically for CS/SPD use.
Disassembly details
“If it isn’t clean it isn’t sterile”— the mantra of every sterile processing technician,” said Lindsay Brown, CRCST, CFER, CCSVP, Clinical Education Manager, Key Surgical. “There are many products on the market that set SPD techs up for success during the cleaning of instrumentation, both manual and mechanical. Cleaning brushes will always be an important tool to help remove bioburden from the inside and outside of surgical instrumentation but it doesn’t stop there!
“Once instruments are cleaned using the correct size brush with the correct bristle type, they are then run through an automatic washer/disinfector,” she adds. “Utilizing the appropriate mix of heat, chemistries and impingement, the instruments have a better chance at getting clean if they are placed in a container that allows that perfect mix to work. Mesh baskets and mesh bags are examples of containment devices that not only protect instruments and keep them from falling off the wash rack but also allow for adequate contact of the detergent. Having the wrong type of tray could impede the wash cycle’s effectiveness. The details matter in order to set instrument trays up for a successful sterilization process.”
Disassembly is an important step in proper reprocessing for all complex instrumentation and keeping small parts and accessories together can be challenging. Key Surgical’s Mesh Bag helps keep small parts, such as endoscope valves, together during reprocessing. Designed to withstand gas plasma, EtO, and high-level disinfectant.
Harmful fumes and odors
“One of the common challenges faced today is long hours in front of the manual cleaning sink on the decontamination side inhaling odors such as blood on instruments, Subtilisin enzymes and cleaning solution fragrances designed to mask smells,” said Alison Shrader, Product Manager, Belimed. “Some staff members are sensitive to the mixture of odors and cleaning solutions causing headaches and respiratory issues; whereas exposure to Subtilisin enzymes, in general, can cause skin irritations and watering eyes.”
“The key to resolving the issue is to remove the irritants or better yet, not introduce them in the first place,” added John Nies, Product Manager, Belimed. “While you can’t remove bloody instruments from the process, looking for and using multi-enzyme cleaning solutions that are fragrance and Subtilisin free yet still effective at breaking down proteins would help eliminate some of the sources of irritation.”
Belimed now offers a full line of Subtilisin and fragrance free detergents that will clear the air in the CS/SPD creating a better work environment for the staff. Belimed Protect is a proprietary triple enzyme (protease, lipase, amylase) formulation that has the cleaning power to remove blood from instruments and targets the fat, proteins and starch present in biosoils, found on surgical instruments.
Instrument complexity
“Instrument complexity and having the correct tools to clean those instruments are some of the biggest challenges I see the SPD staff has to overcome to effectively prepare instrumentation for sterilization,” said Aaron Lieberman, Marketing Manager, Summit Medical. “I am always amazed at the skill level and the knowledge the SPD staff has to disassemble some instruments per manufacturer’s instructions to ensure they are cleaning every possible nook and cranny. But even after the instrument is disassembled, it’s essential they are using the right cleaning tool, or more specifically, the right sized brush. To be successful it’s important to have a variety of brush types with various bristle materials and diameters on hand to meet the cleaning requirements outlined in the IFUs.”
Summit Medical recently launched its InstruSafe Care + Maintenance product line. All of the company’s instrument cleaning products are gentle, yet effective, accommodating even the most unique and delicate tools. Summit Medical’s General Purpose Brushes can be used on a variety of instruments. According to Lieberman, these brushes are strong enough to remove anything the OR throws at them, yet delicate enough to leave equipment
unharmed.
Visual inspection
“One of the biggest challenges CS/SPD staff face when it comes to successful decontamination is visual inspection of the inside of instruments that can be potentially soiled,” said Matthew Smith, Marketing Manager for Healthmark Industries. “There is virtually no way to determine if they are properly cleaned without specialized instruments. There are new innovative products that not only include camera’s and light sources needed to perform a visual inspection, but also software integrated that will allow the user to record and document what they are viewing.”
Healthmark recently introduced the FIS-003 Flexible Inspection Scope to its Prosys Optical Inspection line of products. The FIS-003 features a longer 110cm flexible shaft with graduation marks. Like other flexible inspection scopes, it also includes a distal tip composed of a light source and camera lens at the end of the flexible shaft. Designed for instruments 2.4mm in diameter or larger, inspection is easier on the interior of instruments with small diameters, including many flexible GI endoscopes. The camera and light are powered by the USB connection on a PC.
Workflow efficiency
“Having access to the appropriate disinfection equipment is a common challenge for staff attempting to achieve successful decontamination,” said Jason Simon, Product Manager, Infection Prevention, Skytron. “Today’s more sophisticated medical devices are provided with varying instructions for use (IFU) listing detailed directions on how to process with specific times and temperatures. There are many healthcare facilities that continue to use outdated equipment that is not equipped to meet these challenges or worse, no automated disinfection equipment at all. Some medical device IFU’s provide instructions for proper manual cleaning process but the time permitted to follow these instructions are not realistic.”
The Skytron Sterling P1500 washer-disinfector streamlines and automates the decontamination process — bringing unsurpassed efficiency and flexibility to the sterile processing department. The Sterling P1500 allows for 40 fully customized cycles, each with 10 available wash phases, providing the flexibility required to meet any thermal disinfection need. Intuitive touchscreen controls enable users to add or edit parameters for pre-wash, wash, rinse or dry phases, and ensure that each water and temperature compatible device is cleaned according to manufacturer specifications.
Cleaning efficacy
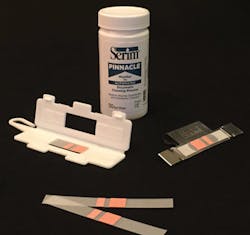
“The continued discovery of outbreaks associated with improperly cleaned medical instruments highlights the importance of regularly testing mechanical cleaning units,” said Greg Sautter, Product Sales Manager, Serim Research Corporation.
The PINNACLE Automated Enzymatic Cleaning Monitor (AEC) from Serim Research tests the cleaning efficacy in both washer-disinfectors and ultrasonic cleaners. This test monitors all aspects of the cleaning process including: enzymatic detergent concentration and activity, wash cycle time, wash cycle temperature and mechanical action of the unit.
Effective drying
“The introduction of potentially harmful contaminants after an ultrasound probe has been properly high-level disinfected is a reality in today’s healthcare environment,” said Mark Leath, President, CS Medical. “A properly dried TEE ultrasound probe, prior to storage, is critical in minimizing the possibility of water-borne bacteria contamination during storage. A wet probe, stored in a plastic sheath or hung freely for drying, can allow airborne contaminates to foster growth of spores and bacteria. Some of the current methods employed for drying after high-level disinfection include; reusable sponge, paper towel, cotton cloth and surgical gauze. These solutions, though effective in the drying of a TEE probe, could introduce outside contaminates. The sponge, if reused, could foster spore and bacteria growth, while the towel, cloth and gauze could contain outside contaminates introduced during the manufacturing or handling process prior to being used.”
CS Medical’s QwikDry removes the current issues associated with other drying methods employed in today’s healthcare facilities. QwikDry is an individually packaged, irradiated cloth with a super absorbent matrix and ultra-smooth texture surface that effectively removes moisture and slides freely over the TEE ultrasound probe shaft. Each cloth is designed for single-use, thus removing the potential for cross-contamination and potential microbiological growth. QwikDry ultrasound probe drying cloths are the next solution for healthcare professionals that are tasked with minimizing healthcare associated infections.
About the Author
Kara Nadeau
Senior Contributing Editor
Kara Nadeau is Sterile Processing Editor for Healthcare Purchasing News.