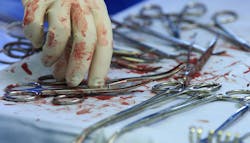
Quality sterile processing starts with IFUs at point of use
The single most important duty and responsibility of everyone involved in sterile processing of reusable medical devices is to protect patients from harm caused by a contaminated device being returned to surgery. This responsibility begins with pre-cleaning the device at the point-of-use and it includes all of the personnel involved throughout the entire decontamination, cleaning and sterilization processes. The manufacturer of the device has an equal responsibility to protect patients from harm by validating the cleaning and sterilization Instructions for Use (IFU) to prove that they actually work to provide a clean, sterile, moisture-free device from every processing cycle.
Unfortunately, many people mistakenly believe that if a device has been sterilized, even though it remains contaminated with organic debris after cleaning, it is safe to use on a patient. Nothing could be further from the truth. Any device that remains contaminated with residual organic debris is not safe to use, even after complete and thorough sterilization. If it’s not clean prior to sterilization, it is absolutely not safe for use with a patient.
Quality sterile processing cannot be accomplished without the thorough and complete cleaning of the device. The cleaning of a reusable medical device is defined as the process of removing organic and inorganic debris from the device and is typically accomplished using water with detergents or enzymatic products that are suitable for use. A device must be thoroughly cleaned before high-level disinfection and sterilization because foreign debris can interfere with the effectiveness of these processes.1
1. The first step in quality sterile processing begins with treating the device at point=of-use. The Association of Surgical Technologists (AST) publication, “Standards of Practice for the Decontamination of Surgical Instruments,” states that, “The cleaning of instruments should begin during the surgical procedure to prevent drying of blood, soil and debris on the surface and within lumens.”
The proper point-of-use treatment of a device starts by placing the contaminated device into water or an approved detergent. This initial step in the process is vital because if soiled organic materials dry or bake onto the device, the removal process becomes much more difficult. Even worse, it can render the disinfection or sterilization process less effective, or even ineffective.2 Reusable medical devices must be presoaked or rinsed in neutral pH water or in a detergent solution immediately after use to prevent blood from drying. A spray detergent may be used in place of water or a detergent solution to keep the devices moist during transport. Once the devices have been treated at the point of use they are ready for transport to the decontamination area.
The AST is not alone in recommending the treatment of devices at the point of use. The AORN recommends pre-cleaning at point-of-use as well.3 Finally, point-of-use treatment is recommended and covered in sections 6.3 thru 6.4 in AAMI ST79 (2017).
2. The next step in quality sterile processing involves the decontamination and cleaning of the device. Manual cleaning is required in those areas that do not have mechanical cleaning units (e.g., ultrasonic cleaners or washer disinfectors). Manual cleaning is also required for fragile or difficult-to-clean instruments (e.g., Kerrison Rongeurs, laparoscopic instruments, etc.). There are two essential components of manual cleaning, friction and fluidics. Friction (e.g., rubbing/scrubbing the soiled area with a brush) is a time-honored and dependable method. Fluidics (i.e., fluids under pressure) is used to remove soil and debris from internal channels, sharp internal corners and dead-spaces.4
The most common types of mechanical or automatic cleaners are ultrasonic cleaners, washer decontaminators, washer-disinfectors and washer-sterilizers. The use of a washer-disinfector requires additional care when loading surgical instruments into the unit. Specifically, hinged instruments must be fully opened to allow thorough and complete contact with the detergent solution. Instruments should not be stacked in washers to ensure proper contact with the detergent solution on all of the surfaces. Finally, all modular/take-apart instruments should be disassembled as per the manufacturer’s IFU.5
Ultrasonic cleaners remove organic soils and bioburden by cavitation and implosion. This involves oscillating the liquid cleaning solution 12,000 to as much as 16,000 times per second, which generates powerful waves of sonic energy. These oscillating waves of liquid energy break up the adhesion that holds the contaminants to the surfaces of the devices.
Sterile Processing can ensure that devices are completely clean after processing and prior to the sterilization cycle. Conduct a verification test to help ensure that the device is clean and ready for further processing. These tests include protein tests and adenosine triphosphate (ATP) bioluminescence tests, both of which test for residual soils. Further, other options include reagent tests that test for protein and hemoglobin, and another 3-in-1 test for lumen devices that test for blood, protein and carbohydrates, all at the same time.6
Also consider using devices that have had their cleaning and sterilization IFUs validated by an independent testing laboratory using AAMI and FDA ”worst case” cleaning validation testing protocols. Without a validated IFU, processing personnel can be doing everything right, according to the manufacturer’s non-validated IFU, and still not have the assurance of sending clean, sterile, moisture-free devices back to surgery and potentially causing patient harm.
Hence, to ensure quality sterile processing in your facility you must demand that device manufacturers provide a copy of their validated IFU to prove that they actually work. “When asking a device manufacturer for a copy of their validated IFU, you need to be sure the manufacturer understands you are asking for more than just cleaning and sterilization instructions. The manufacturer must provide you with the independent laboratory validation test report that proves that their IFUs actually work for cleaning and sterilizing the device. If a device manufacturer can’t – or won’t – provide you with cleaning and sterilization IFUs that have been independently validated using AAMI and FDA testing protocols, then you need to look for another device manufacturer who can and will.”7
Given the importance of validated IFUs for patient safety, what is a good information source to learn more about this vital subject? Outside of HPN there’s a wealth of helpful information online about IFUs, such as #IFUcan. Created by a group of independent, veteran healthcare executives, advocates and experts in sterile processing and distribution (SPD), #IFUcan explores the use of validated IFUs to improve sterile processing quality and reduce the risk of patient harm caused by a device that remains contaminated after processing. Visit https://www.linkedin.com/showcase/68681202/admin/ for more details.
1 https://www.cdc.gov/infectioncontrol/guidelines/disinfection/cleaning.html2 Ibid, “CDC”3 https://aornguidelines.org/tool/content?gbosid=3968704 Reichert M. Preparation of supplies for terminal sterilization. In: Reichert M, Young JH, eds. Sterilization technology for the health care facility. Gaithersburg, MD: Aspen Publication, 1997:36-50.5 Ibid, “CDC”6 Ibid, “CDC”7 Validating IFUs – Distinguishing Fact from Fiction, HPN, November 2019
About the Author