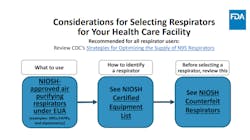
FDA reissues NIOSH-approved EUA to remove decontaminated or expired disposable respirators
On July 12, 2021, the U.S. Food and Drug Administration (FDA) revised and reissued the NIOSH-approved Emergency Use Authorization (EUA) so the scope of the products authorized under that EUA is in alignment with the actions taken on June 30, 2021, as well as FDA’s related announcements of April 9 and May 27.
On June 30, 2021, the FDA revoked EUAs for decontamination and bioburden reduction systems. These devices are no longer authorized for use by healthcare personnel in healthcare settings.
The FDA also no longer believes that facilities should be using crisis capacity strategies. As a result, the NIOSH-approved EUA no longer authorizes the use of NIOSH-approved respirators that are expired or have been processed by any previously authorized decontamination or bioburden reduction system.
Also on June 30, 2021, the FDA issued an updated Letter to Healthcare Personnel and Facilities: FDA No Longer Authorizes Use of Non-NIOSH-Approved or Decontaminated Disposable Respirators, to inform the healthcare community about these revocations.