Removing biofilms; using indicators in sterilization assurance
Submit your questions: email: [email protected]
Q
Over the past few years I have heard a lot about biofilms and how bad they are in sterile processing, but I have to admit I really don’t understand what they are or why they are so bad for us. I have worked in sterile processing for over 20 years and now we have biofilms to worry about. Why is that and what’s next?
A
Over the years we have made many scientific discoveries and advances and have developed lots of new technologies that have changed and improved the way we do things. While biofilms have existed forever, through more advanced study, we have learned more about them and how they can present a significant challenge to effective cleaning and sterilization.
So, what is a biofilm? Well, just as the name implies it is a biological film. This film, or slime, often consists of diverse colonies of tiny microorganisms bound together in a “matrix,” or a thick, sticky substance. This “matrix” consists of both living and dead cells as well as polysacharides or carbohydrates.
Biofilms often are not detectable by touch or visible to the naked eye. However, in extreme cases and with multiplication, biofilms may become more visible and become slippery to touch. Biofilms form when single microorganisms attach to a hydrated surface and undergo a “lifestyle switch,” giving up their life as a single cell to live on a surface in an adhesive cell formation (matrix) with other microorganisms.
Q
I am a second-year nursing student and have been working part time in sterile processing. I recently listened to a podcast on sterilization quality control. I believe I heard one of the speakers say that neither a chemical indicator nor biological indicator ensure that the contents of a sterile package are sterile. I, as well as my co-workers, found his comment very confusing and contradictory. How can a “sterile package” not contain sterile items? If that were so, it would not be a sterile package. Further, why do we use these indicators and tests if they cannot ensure the items are sterile?
A
The sucess of a sterilization process is dependent on many factors. Foremost, the sterilizer must be functioning properly, and the sterilization cycle must meet the required conditions and sterilization parameters of proper steam, temperature, pressure, and time; together these elements assure the effectiveness of this sterilization process.
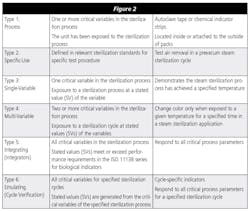
There are six different types of chemical indicators – each type monitors or measures one or more specific parameters or critical variables of the sterilization process (see figure 2). A biological indicator (BI) is a challenge test, which consists of vial that contains active spores. The BI is placed within the sterilizer and/or in containers and packages to be sterilized. Post sterilization and incubation, the BIs are evaluated to verify that the spores were adequately exposed to the required sterilization parameters and destroyed.
Chemical and biological indicators tell us that the sterilization parameters were achieved within the sterilization cycle, thus providing confidence that the conditions were adequate to sterilize all contents in the sterilizer, pending the efficacy of the human element performance, e.g. cleaning, following the device manufacturer’s IFU, packaging, and loading the sterilizer. If there are failures in staff performance, it could be possible to have an unsterile item next to a passed BI in a sterile container or package.
About the Author

Ray Taurasi
Ray Taurasi is Principal, Healthcare CS Solutions. His healthcare career spans over five decades as an Administrator, Educator, Technologist and Consultant. He is a member of AORN, SGNA, AAMI and a past president of IAHCSMM. Taurasi has been a faculty member of numerous colleges teaching in the divisions of business administration, nursing, and health sciences. He is the author of numerous articles and textbook chapters; he is a frequent speaker at national and international healthcare conferences.
Note to readers from Ray Taurasi - In 2021, my life’s career path will transition to one of new opportunities and adventures. As a result, after nearly 19 years and 225 CS Solution columns, this edition will be my last.
“All changes, even the most longed for, have their melancholy; for what we leave behind us is a part of ourselves; we must die to one life before we can enter another.”– Anatole France
I wish you and your loved ones a healthy and joyful holiday season and a beautiful New Year! God Speed, Ray
https://www.facebook.com/pages/category/ Local-Business/Healthcare-CS-Solutions-128857 3061153887/ • email: [email protected]