Addressing supply shortages with standard identifiers
Using standard device identifiers can deliver multiple benefits, from touchless orders to more accurate clinical supply documentation. But what about addressing the supply shortages that continue to plague hospitals? Standard identifiers, in combination with classification codes and descriptions, can help healthcare delivery organizations find acceptable alternatives in the event of shortages, while more widespread use across healthcare can also help ensure products are where they are needed most. In this month’s column, we will explore what is possible if a single health system, or even the entire country, used standard product identifiers like GS1 Global Trade Item Numbers (GTINs) to identify products.
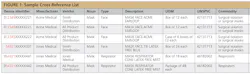
The GTIN is one of the codes compliant with the U.S. FDA’s UDI rule, which requires manufacturers to assign a unique device identifier (UDI-DI) to their products at each packaging level. For example, the same product packaged in a box of 12 would have one UDI-DI, while the same product packaged in a case of 6 boxes of 12 each would have a different UDI-DI (illustrated in Figure 1 as the different last digit in the device identifier column and in the UOM column).
How creating a cross-reference table supports addressing vendor backorders
Figure 1 is a cross-reference table (simplified for demonstration purposes) that shows how a hospital could find alternative vendors or products using GTINs as the UDI-DI.
Let’s say your hospital normally buys a mask manufactured by Acme Medical from Smith Distribution. If Smith Distribution is not able to supply the mask, you could use the GTIN in the first column to determine that another vendor, All Products Distribution, sells the exact same mask, packaged either as a box of 12 each, or as a case of 6 boxes of 12 each. The similar numbers in red represent a company prefix associated with the manufacturer in the GS1 system.
But what if Acme Medical cannot supply the masks to either distributor? In this case, your hospital could use the combination of the UNSPSC classification code (grouping similar products) and the description to find a suitable substitute, this time made by a different manufacturer but sold by your usual vendor, Smith Distribution. The different company prefix represents the different manufacturer.
Now, let’s consider what happens when you are not able to procure a product, this time a respirator, directly from the manufacturer. By looking at the last two rows in Figure 1, your hospital determines that the same product (but at a different packaging level) is sold via distribution from All Products. Note the device identifier has the same company prefix but a different last digit in the device identifier column and the different UOM.
Using standard identifiers to address widespread shortages
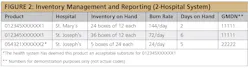
Figure 2 depicts the potential value to a single, multi-hospital system, as well to the nation as a whole, in the event of widespread shortages like we experienced with personal protective equipment (PPE) early in the pandemic. During those shortages, every hospital system reported their PPE inventory levels and burn rates. If a health system uses UDI-DIs across hospitals, it could see the respective inventory and consumption levels, both for the same product and ones that it has deemed acceptable substitutes, and as such, could move product to where the need is greatest.
Now consider if all hospitals not only used UDI-DIs in those reports, but also all of healthcare, including the Strategic National Stockpile (SNS) and both PPE manufacturers and distributors. In this case, we would have had a much better understanding of where the need was greatest as the pandemic hit different parts of the country harder at different times. Further, with use of the Global Medical Device Nomenclature (GMDN), a code based on regulatory approval based on use and available in the FDA’s Global UDI Database, the SNS could determine functional equivalency in emergency situations. As such, we would have had much better data with which to prioritize deliveries to support needs-based allocation.
While both of these illustrations are focused on emergency situations, the examples also support healthcare’s long desire to implement demand planning at scale. See the September 2021 issue of Standard Practices. In the spirit of never let a good crisis go to waste, now is the time to leverage the UDI rule, which has made UDI-DIs available on most medical products for emergency planning, but also to support our ongoing need for better supply and demand matching to support healthcare delivery.
About the Author

Karen Conway
CEO, Value Works
Karen Conway, CEO, ValueWorks
Karen Conway applies her knowledge of supply chain operations and systems thinking to align data and processes to improve health outcomes and the performance of organizations upon which an effective healthcare system depends. After retiring in 2024 from GHX, where she served as Vice President of Healthcare Value, Conway established ValueWorks to advance the role of supply chain to achieve a value-based healthcare system that optimizes the cost and quality of care, while improving both equity and sustainability in care delivery. Conway is former national chair of AHRMM, the supply chain association for the American Hospital Association, and an honorary member of the Health Care Supplies Association in the UK.