May 2021 Product Picks
Ergonomic mat support
An anti-fatigue mat that helps reduce fatigue and discomfort for healthcare workers standing for long hours in reprocessing areas has been added to the personal protection equipment (PPE) product line of Healthmark Industries. Manufactured from energy-return polyurethane foam, the black 0.75-Inch thick mat has a stain and puncture-resistant surface to prevent absorption of dirt and water, a beveled edge at 18 degrees to reduce tripping potential, matte textured top surface to provide friction and anti-microbial properties for added protection. It is equipped with a high-traction bottom surface certified by the National Floor Safety Institute. Sizes include 20 x 32 inches, 24 x 36 inches, and 20 x 48 inches.
Bioprocessing single-use parts
Components for the design, development and manufacture of single-use systems (SUS) developed specifically for the needs of the bioprocess industry is a new extension to the product portfolio of Qosina. The company is a global supplier of OEM single-use components to the medical and pharmaceutical industries. Its single-use bioprocess components are supported by regulatory documentation and backed by the company’s assurance of supply.
Therapeutics and medical business facility
A new facility in Westborough, MA has been opened to serve as the global headquarters and an R&D hub for Olympus Corporation’s Therapeutic Solutions Division (TSD) and its medical business in the United States. The 150,000-square-foot Olympus Westborough will accommodate the U.S. TSD employees and those formerly reporting to Image Stream Medical (ISM) facility in Littleton, MA, with the exception of the manufacturing employees who will remain in Littleton. The facility will include hands-on learning in a wetlab setting in person or virtually through telecollaboration and a showroom for in-person or virtual OR demonstrations.
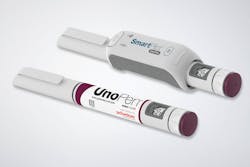
Insulin injection smart technology
An electronic communication interface that aids in insulin injection by pens for patients with diabetes has been developed by Schreiner MediPharm. The NFC-Label has an integrated NFC chip that combines with the UnoPen, a disposable pen with variable dose setting offered by Ypsomed, and the SmartPilot. With the label, the drug can automatically be identified, authenticated and checked in terms of its expiration date. The time and date of the injection and the delivered dose are tracked and transmitted to the patient’s smartphone app via Bluetooth. Patients are interactively guided through the injection process, assisted in correctly using the pen or informed about inconsistencies. Sensor technology installed in the SmartPilot digitally connects the UnoPen to temperature monitoring, which issues a warning if the pen was exposed to critical temperatures. Schreiner MediPharm